Two members of our research group are listed as co-authors in one of the Hungarian Academy of Sciences’ featured publications.
Tamás Holczbauer co-authord the article: Reimagining o-Carborane-Based Fluorophores: Charge-Transfer from Boron Substituents Triggers Aggregation- and Crystallization-Induced Emission published in Angew. Chem. Int. Ed. 2026 by
Balázs Szathmári, Yanhong Gao, Dániel Buzsáki, Dániel Zámbó, Tamás Holczbauer, Antal Udvardy, Pál Szabó, Júlia Kertész, Dóra Hessz and Zsolt Kelemen. (https://doi.org/10.1002/anie.202521741)
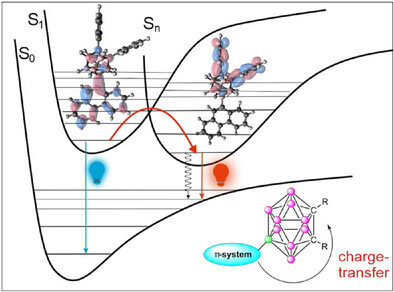
o-Carborane-based fluorophores are widely recognized for their aggregation-induced and crystallization-induced emission (AIE and CIE) properties, making them valuable building blocks for materials used in fluorescence applications such as bioimaging or optoelectronics. Until now, observing AIE/CIE required the fluorophore to be attached via the carbon atom of the carborane, inherently limiting further functionalization possibilities. Herein, we report for the first time that boron-substituted carboranes (substituent is at position 9) exhibit AIE/CIE activity, which originates from photoexcitation-induced electron-transfer from the substituents linked to the carborane cage via its boron vertices. Furthermore, the solid state emission can be tailored by the proper selection of the substituents. This finding not only significantly broadens the scope of AIE-active compounds but also offers new design strategies for the development of luminescent carborane-based materials.

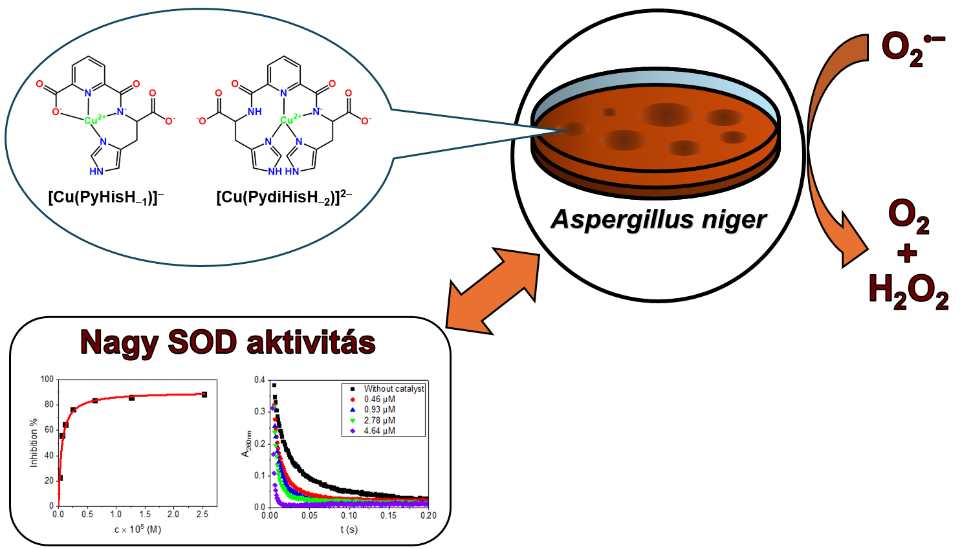
Nóra V. May co-authored the article: Highly active superoxide dismutase mimic: pyridine carboxamide-based copper(ii) complexes in the Inorganic Chemistry Frontiers, 2025 by Róbert Diószegi, Andrea Guidetti, Norbert Ág, Ilenia Serra, Dóra Szalóki, Dóra Bonczidai-Kelemen, Nóra V. May, Erzsébet Fekete, Levente Karaffa, István Fábián, Sabine Van Doorslaer and Norbert Lihi. (https://doi.org/10.1039/D5QI01687H)
Pyridine-based ligands functionalized with one (PyHis) or two histidine (PydiHis) moieties were synthesized, and their copper(II) complexes were studied as novel antioxidant systems. The complexes were characterized by pH-potentiometric, spectroscopic (UV-vis, circular dichroism, continuous-wave and pulsed electron paramagnetic spectroscopy (EPR)), computational (DFT) and X-ray diffraction methods. The complexes show high thermodynamic stability. The PyHis complex possesses a (COO−,Npy,N−,NIm) donor set, while PydiHis binds copper(II) via a (NIm,Npy,N−,NIm) donor set at physiological pH. The PyHis and PydiHis complexes of copper(II) exhibit negligible (IC50 = 147 ± 15 μM) and outstanding (IC50 = 0.64 ± 0.07 μM) superoxide dismutase (SOD) activity, respectively. The noted difference is attributed to the structural flexibility of the latter complex as confirmed by pulsed EPR spectroscopy. The antioxidant activity of the copper(II) complexes was also tested in vivo by monitoring their effects on the mycelial growth of Aspergillus niger under oxidative stress. Both complexes were able to protect the cells under these conditions; however, the copper(II) complex of PydiHis was more effective in accordance with its excellent SOD activity. Consequently, the copper(II) complex of PydiHis stands out as an efficient, low-molecular-weight SOD mimic combining high catalytic activity with proven biological efficacy under oxidative stress conditions, making it a promising candidate for medical applications.